NCG EQAS
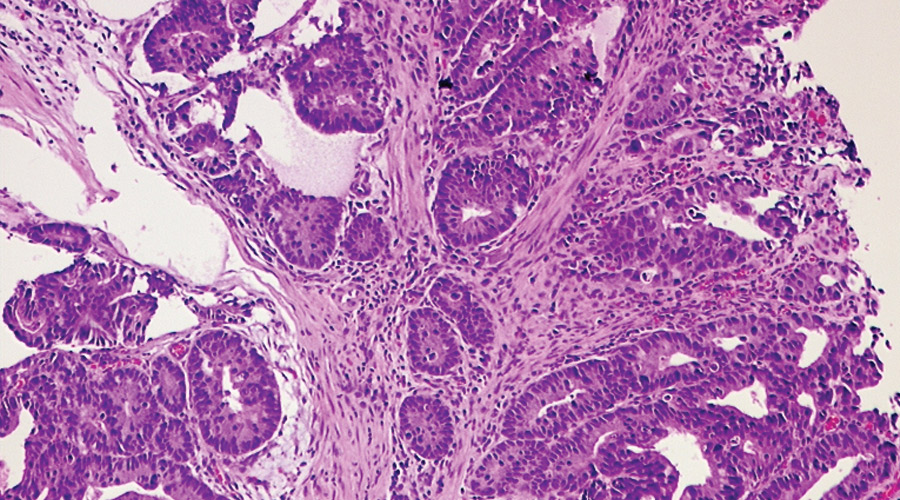
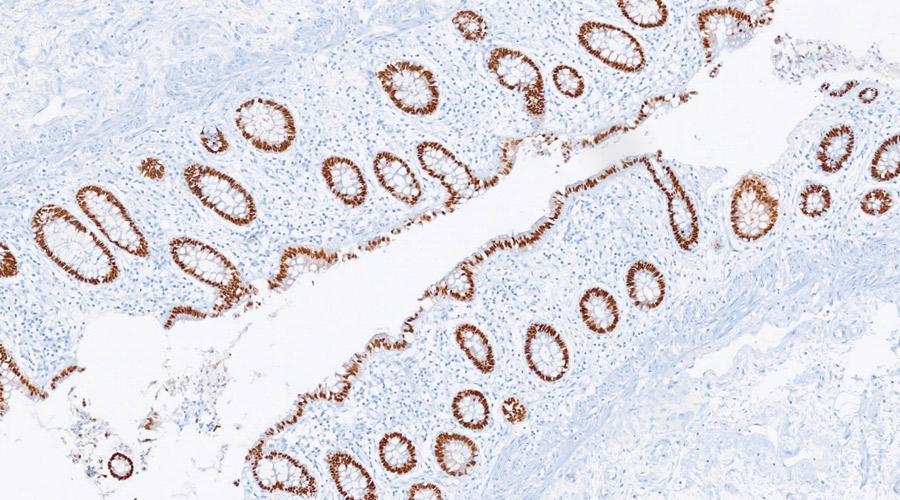
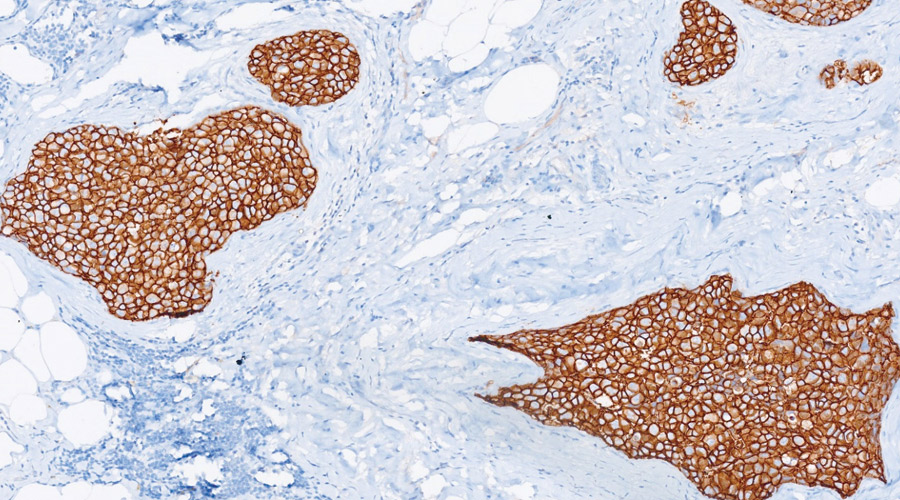
Quality control is the basic step in pathology practice in order to ensure accurate patient diagnosis. With the widespread availability of targeted therapeutic options for various cancers, prognostic and therapeutic biomarkers have gained the forefront in guiding patient treatment and outcome. For maximum patient benefit all test results should be reproducible. This requires a consensus form of testing and reporting across all the pathology laboratories. With this vision in mind, we have started External Quality assurance Scheme (EQAS) for surgical pathology under the realm of National Cancer Grid.
History
The National Cancer Grid (NCG) was formed in August 2012 with the mandate of linking cancer centres across India. The initiative, which originally had 14 cancer centres, has rapidly grown now to include 142 major cancer centres, research institutes, patient groups and philanthropic organizations across the country and is amongst the largest cancer networks in the globe. Funded by the Government of India through the Department of Atomic Energy, the NCG has the primary mandate of working towards uniform standards of care across India by adopting evidence-based management guidelines, which are implementable across these centres.
Aim
Surgical Pathology and Oncology practices in India are very heterogeneous and often dictated by patient’s affordability. Quality should not be dependent on economics and hence NCGEQAS was started under the aegis of NCG to work towards a common practice for all. The NCGEQAS Program is funded by NCG.
Policy
The program provides access to all the NCG registered centers for free while others are nominally charged. Education is important aspect of each Run report and we take into account the lack of infrastructure in many centers while issuing final reports. All the protocols are as per the standardization carried out at our laboratory. This program is presently being run by and in Tata Memorial Hospital.